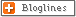The Major Body Buffer Systems
Site Buffer System Comment
1. ISF
a. Bicarbonate For metabolic acids
b. Phosphate Not important because concentration too low
c. Protein Not important because concentration too low
2. Blood
a. Bicarbonate Important for metabolic acids
b. Hemoglobin Important for carbon dioxide
c. Plasma protein Minor buffer
d. Phosphate Concentration too low
3. ICF
a. Proteins Important buffer
b. Phosphates Important buffer
4.Urine
a. Phosphate Responsible for most of 'Titratable Acidity'
b. Ammonia Important - formation of NH4+
5. Bone
a. Ca carbonate In prolonged metabolic acidosis
The other buffer systems in the blood are the protein and phosphate buffer systems.
These are the only blood buffer systems capable of buffering respiratory acid-base disturbances as the bicarbonate system is ineffective in buffering changes in H+ produced by itself. The phosphate buffer system is NOT an important blood buffer as its concentration is too lowThe concentration of phosphate in the blood is so low that it is quantitatively unimportant. Phosphates are important buffers intracellularly and in urine where their concentration is higher.
Haemoglobin is an important blood buffer particularly for buffering CO2
Protein buffers in blood include haemoglobin (150g/l) and plasma proteins (70g/l). Buffering is by the imidazole group of the histidine residues which has a pKa of about 6.8. This is suitable for effective buffering at physiological pH. Haemoglobin is quantitatively about 6 times more important then the plasma proteins as it is present in about twice the concentration and contains about three times the number of histidine residues per molecule.
The carbonate and phosphate salts in bone act as a long term supply of buffer especially during prolonged metabolic acidosis. The important role of bone buffers is often omitted from discussions of acid-base physiology.
Bone is the major CO2 reservoir in the body and contains carbonate and bicarbonate equivalent to 5 moles of CO2 out of a total body CO2 store of 6 moles. (Compare this with the basal daily CO2 production of 12 moles/day)
CO2 in bone is in two forms: bicarbonate (HCO3-) and carbonate (CO3-2). The bicarbonate makes up a readily exchangeable pool because it is present in the bone water which makes up the ‘hydration shell’ around each of the hydroxyapatite crystals. The carbonate is present in the crystals and its release requires dissolution of the crystals. This is a much slower process but the amounts of buffer involved are much larger.
How does bone act as a buffer?
Two processes are involved:
• Ionic exchange
• Dissolution of bone crystal
Bone can take up H+ in exchange for Ca++, Na+ and K+ (ionic exchange) or release of HCO3-, CO3- or HPO4-2. In acute metabolic acidosis uptake of H+ by bone in exchange for Na+ and K+ is involved in buffering as this can occur rapidly without any bone breakdown. A part of the so called ‘intracellular buffering’ of acute metabolic disorders may represent some of this acute buffering by bone. In chronic metabolic acidosis, the major buffering mechanism by far is release of calcium carbonate from bone. The mechanism by which this dissolution of bone crystal occurs involves two processes:
• direct physicochemical breakdown of crystals in response to [H+]
• osteoclastic reabsorption of bone.
The involvement of these processes in buffering is independent of parathyroid hormone. Intracellular acidosis in osteoclasts results in a decrease in intracellular Ca++ and this stimulates these cells.
Bone is probably involved in providing some buffering for all acid-base disturbances. Little experimental evidence is available for respiratory disorders. Most research has been concerned with chronic metabolic acidoses as these conditions are associated with significant loss of bone mineral (osteomalacia, osteoporosis). In terms of duration only two types of metabolic acidosis are long-lasting enough to be associated with loss of bone mineral: renal tubular acidosis (RTA) and uraemic acidosis. Bone is an important buffer in these two conditions.
In uraemia, additional factors are more significant in causing the renal osteodystrophy as the loss of bone mineral cannot be explained by the acidosis alone. Changes in vitamin D metabolism, phosphate metabolism and secondary hyperparathyroidism are more important than the acidosis in causing loss of bone mineral in uraemic patients. The loss of bone mineral due to these other factors releases substantial amounts of buffer.
Buffering Capacity
Buffer solution is able to retain almost constant pH when small amount of acid/base is added. Quantitative measure of this resistance to pH changes is called buffer capacity. Buffer capacity can be defined in many ways. You may find it defined as "maximum amount of either strong acid or strong base that can be added before a significant change in the pH will occur". This definition instead of explaining anything - raises a question "what is a significant change?" - sometimes even change of 1 unit doesn't matter too much, sometimes - especially in biological systems - 0.1 unit change is a lot. Buffer capacity can be also defined as quantity of strong acid or base that must be added to change the pH of one liter of solution by one pH unit. Such definition - although have its practical applications - gives different values of buffer capacity for acid addition and for base addition (unless buffer is equimolar and its pH=pKa). This contradicts intuition for a given buffer solution its resistance should be identical regardless of whether acid or base is added. Where n is number of equivalents of added strong base. Note that addition of n moles of acid will change pH by exactly the same value but in opposite direction.
sources :